Blog post
Research in the context of global health emergencies: writing a background paper for the Nuffield Council on Bioethics.
This post was written by Dr Agomoni Ganguli Mitra, Research Associate (Liminal Spaces Project) and Teaching Fellow, Mason Institute, School of Law, University of Edinburgh and Dr Nayha Sethi, Research Fellow (Liminal Spaces Project) and Deputy Director, Mason Institute, School of Law, University of Edinburgh. It first appeared on The Motley Coat - a blog of the Mason Institute
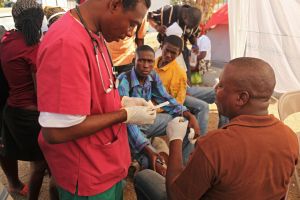
In 2016, in the wake of the Ebola and the Zika outbreaks, the international community’s response to global health emergencies and their associated moral, regulatory and political implications once again came under intense scrutiny. For the bioethics and governance communities these were real challenges, forcing us to revisit some of the most difficult ethical questions around pandemics, such as health inequality, weak health system and access to care, and reproductive health. The challenges were all the greater for having to address them within the global (heterogenous) regulatory systems. The response to Ebola for example, developing at the intersection between humanitarian response, care, and research activities - each with their values and goals - brought into question well established ethical norms and practices. It was against this background that the Nuffield Council on Bioethics launched a call to commission research on research in the context of global health emergencies.
We were fortunate to win the commission to take on this work. The brief was clear: we were to produce a background paper on the ethical and governance issues raised by research in the context of global health emergencies. The result was a challenging, but extremely interesting experience for both of us. As newcomers to the topic, there were unexpected obstacles. For example, we spent quite a few hours questioning what might, or might not count as a global health emergency. We also faced obstacles many academics will be familiar with: how to reduce a wealth of literature (relevant regulatory and governance tools, laws, partnerships, practices, actors, cases, practices and lessons-learned) into a short, comprehensive document that would be accessible, and of use to the Council.
In the end, however, teasing out some of the conflicts was one of the most interesting exercises. For example, in the particular context of infectious diseases, how do we reconcile the ‘moral obligation to learn as much as possible, as quickly as possible’ (WHO 2016) with the seemingly equally important norm of not sacrificing care for the sake of conducting research? Perhaps unsurprisingly, then, our concluding section was organised around various sets of tensions:
- Tensions between ‘response’ and ‘research’: for example, how can care, research and innovation co-exist efficiently and ethically?
- Tensions within research: for example to what extent can we develop anticipatory ethical and regulatory frameworks for research during epidemics, in a context where oversight and governance need to be particularly sensitive and responsive to evolving and emerging situations?
- Tensions arising in priority-setting and collaboration: for example, when engaging local, regional and global communities, how do we ensure that all voices are heard and differing priorities are taken into account?
It is not often that academics witness their work being put to immediate practical use. In this case, our background paper served to inform a workshop at the Nuffield Council on Bioethics, to which we were also invited. The event brought togetherscientists, physicians, responders, regulators, ethicists and other stakeholders to discuss some of the issues arising from the paper. We have certainly been inspired to think in-depth about some of the issues we encountered while writing. We are grateful to the Council for providing us with the opportunity to do so, and we look forward to continuing to contribute to this important field.
The background paper can be accessed here.

Comments (0)
Join the conversation