Medical implants
Policy Briefing
Published 19/06/2019
Efficacy and safety of medical implants
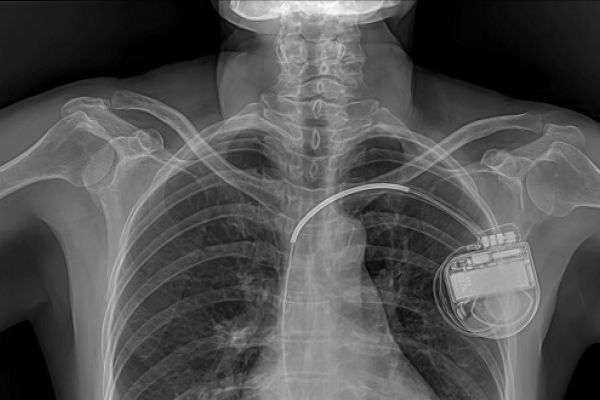
Medical implants can be life-saving. For example, a pacemaker or cardiac defibrillator can prevent life threatening malfunctioning of the heart in at-risk patients. Implants can also restore mobility and improve quality of life. They can help to cut the costs of ill-health by reducing the need for regular treatment or enabling people to return to work. Remote monitoring using active implants can mean fewer visits to hospital are needed, potentially freeing up patient and staff time.
However, implants can pose physical risks arising from surgery, including the possibility that the body might reject or react to the implant. There might also be psychological effects. Some implants, such as deep brain stimulation devices, are associated with effects on mood and behaviour, with possible wider effects on family and social networks. Implantable cardioverter defibrillators issue electric shocks which can be distressing to the patient and have been associated with depression, anxiety disorders, and post-traumatic stress. The UK Government recently launched a review into the safety of medicines and medical devices (see Box 2).
Evidence gathering and clinical trials
Some features of medical implants create challenges for assessing their efficacy and safety while ensuring timely access for patients. In clinical trials of medicines, the medicine can be given in small doses initially and the trial can be stopped at any time. In contrast, medical implants cannot be gradually introduced and once implanted they can be difficult or risky to remove. How well an implant works might also depend on other factors, such as the selection of patients, and the skill and experience of the surgeon. Because implants are often designed to stay in the body for many years, the timeframe for fully testing their lifetime safety and efficacy would be much longer than for medicines.
Technology assessment and guidance
The National Institute for Health and Care Excellence (NICE) can assess the safety and cost-effectiveness of medical implants where they are relevant to NHS guidance on the management of particular conditions. NICE sometimes issues separate guidance on single technologies or interventional procedures, which could involve medical implants, if they offer plausible additional benefits to patients and the healthcare system. The UK Government has committed funding to increase the number of technology assessments carried out by NICE by 2020.
Post-market surveillance
Once an implant has been certified, it is the manufacturer’s responsibility to collect data relating to safety and report adverse incidents to the competent authorities. Under the new EU Regulation, manufacturers must upload information relating to some aspects of their post-market surveillance for each device to the EUDAMED database. However, these data will not be available to the public or to clinicians.
Registries can play an important role in monitoring the safety of implants. For example, the National Joint Registry (NJR) provides early warning for patient safety issues associated with joint replacements and a means of re-contacting patients if issues arise.*NICE has recommended that a national registry of surgery for urinary incontinence and pelvic organ prolapse should be established, which would monitor all uses of vaginal mesh. The Royal College of Surgeons has recommended the establishment of a UK-wide registry to track all new interventional procedures and implants, with independent oversight and review. During Parliamentary debate in February 2019, it was stated that the Government is considering establishing a national medical devices registry.
However, registries are expensive to run and their cost-effectiveness can be difficult to demonstrate. Joining up data from different sources is another approach to monitoring the safety and efficacy of medical implants. Current plans to join up digital systems across the NHS could offer new ways to capture information about the performance and safety of implants.
Box 2. The independent medicines and medical devices safety review
In February 2018, the UK Secretary of State for Health and Social Care announced an independent review into the safety of medicines and medical devices, triggered by concerns about the effects of three medical interventions including vaginal mesh. Mesh was used to treat urinary incontinence in women, but increasing numbers of women reported complications including pain, infection, and mobility problems. The review may make recommendations about how the healthcare system can improve its response to concerns raised about medical devices in the future. The review is expected to conclude in 2019.
* For example, the recall of the DePuy hip implants was in part prompted by data from the NJR showing a higher than expected number of patients requiring revision surgery. National Joint Registry (2017) About the NJR; National Joint Registry (no date) Statement regarding metal-on-metal implants.
Share